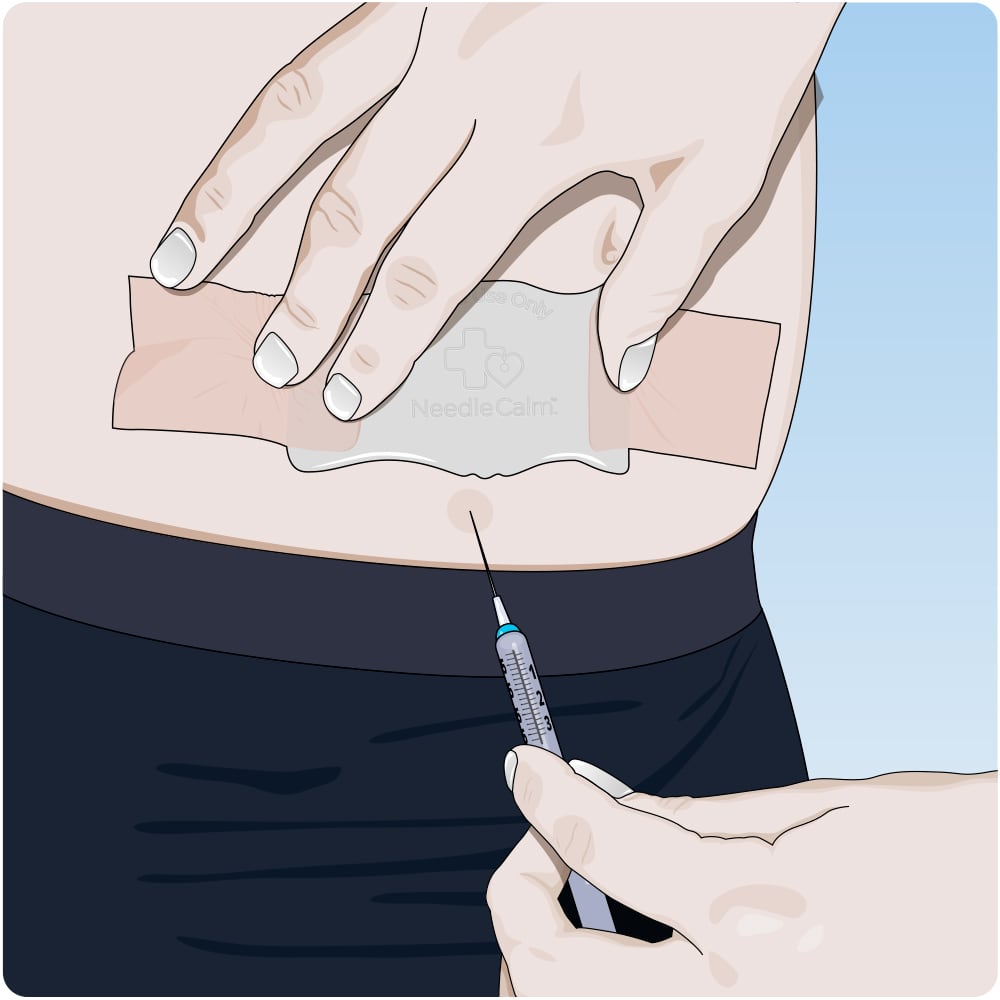
Needle Desensitising Device

-
2021
-
Product
Medical and Scientific
Designed By:
NeedleCalm is a single use adhesive pad with biocompatible medical grade silicone. This Australian designed product was created to promote comfort and minimise the perception of pain associated with injections, immunisations, venepunctures and catheterisation procedures.