Gibco CTS Rotea Counterflow Centrifugation System

-
2021
-
Engineering
Designed By:
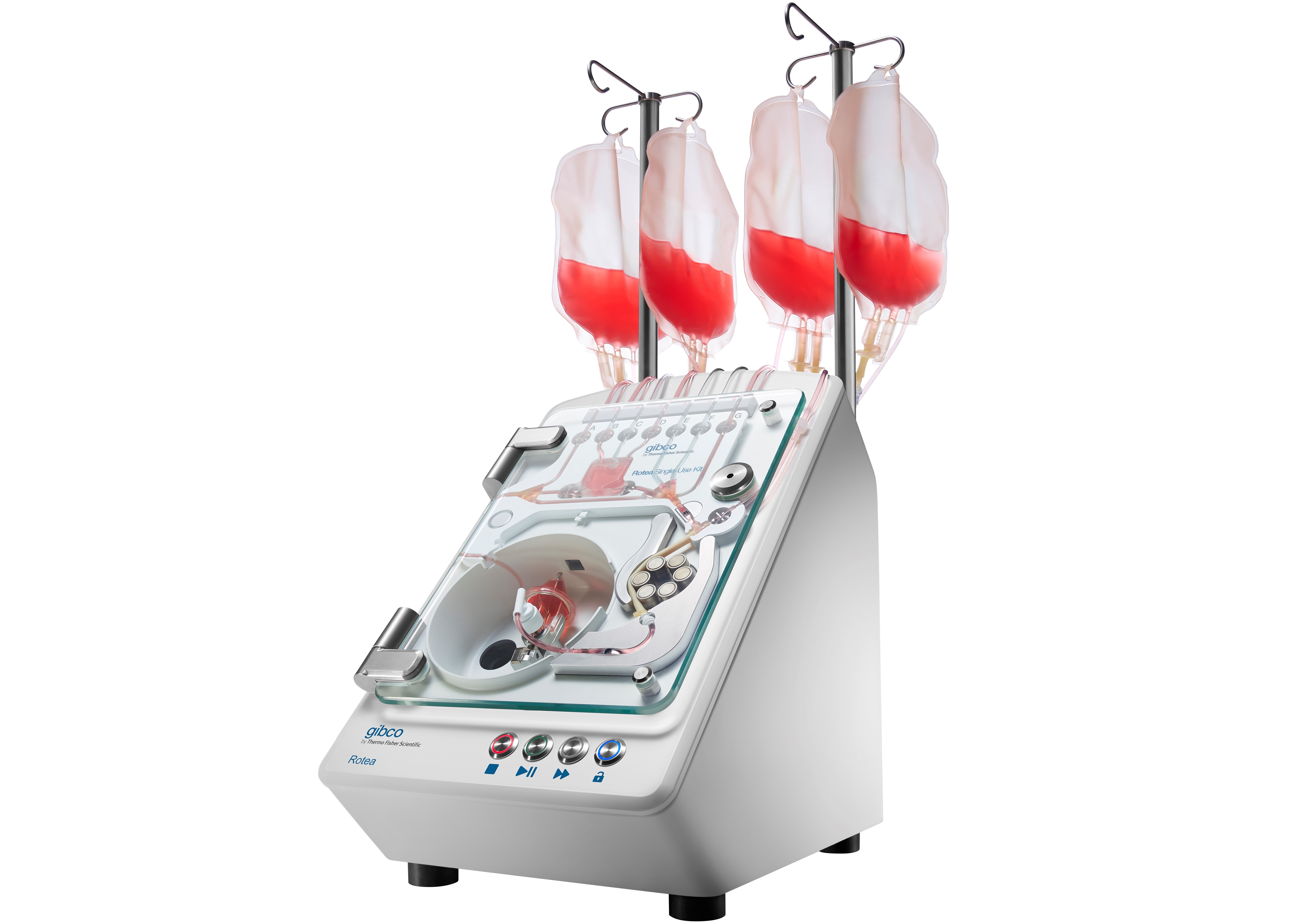
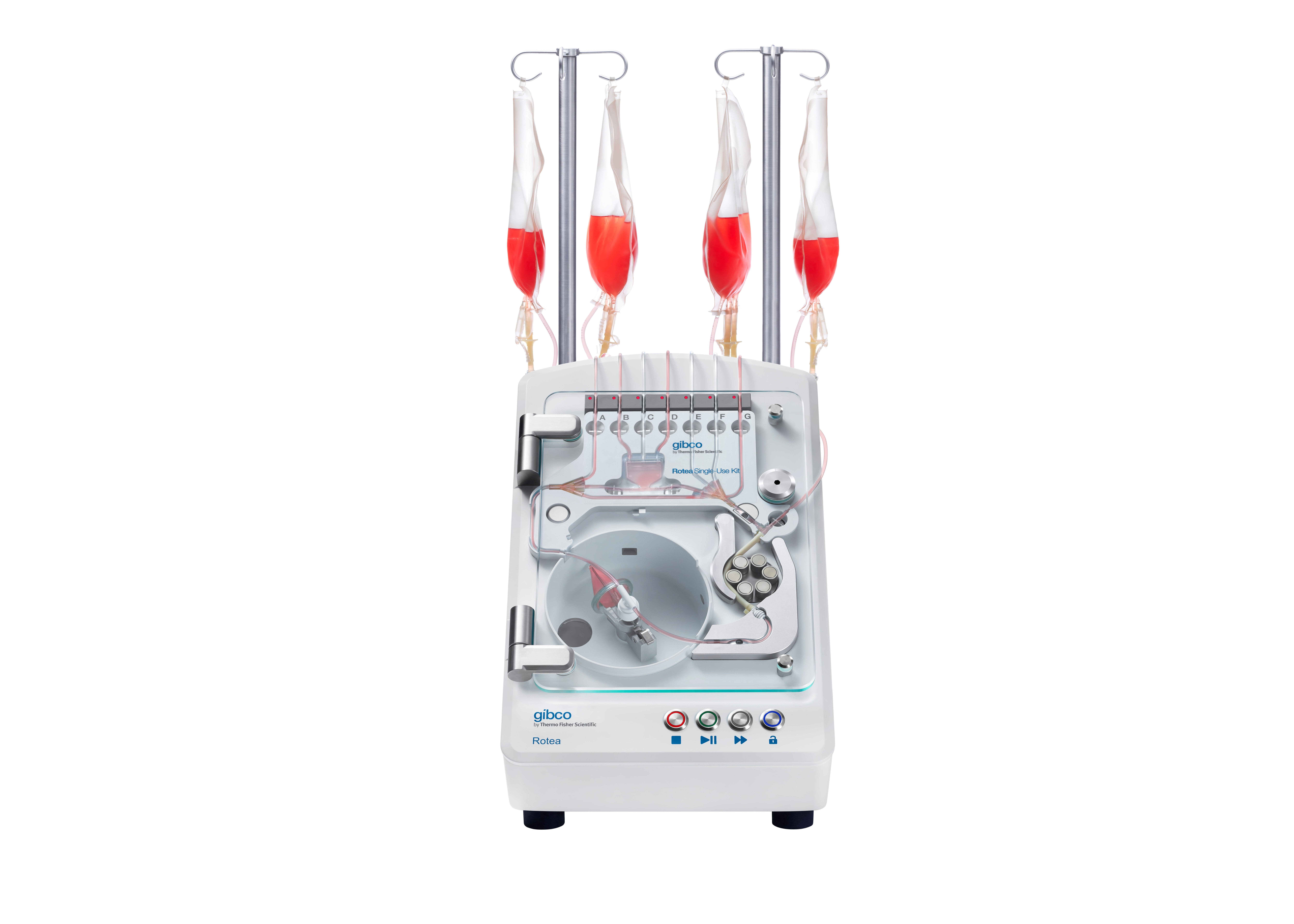
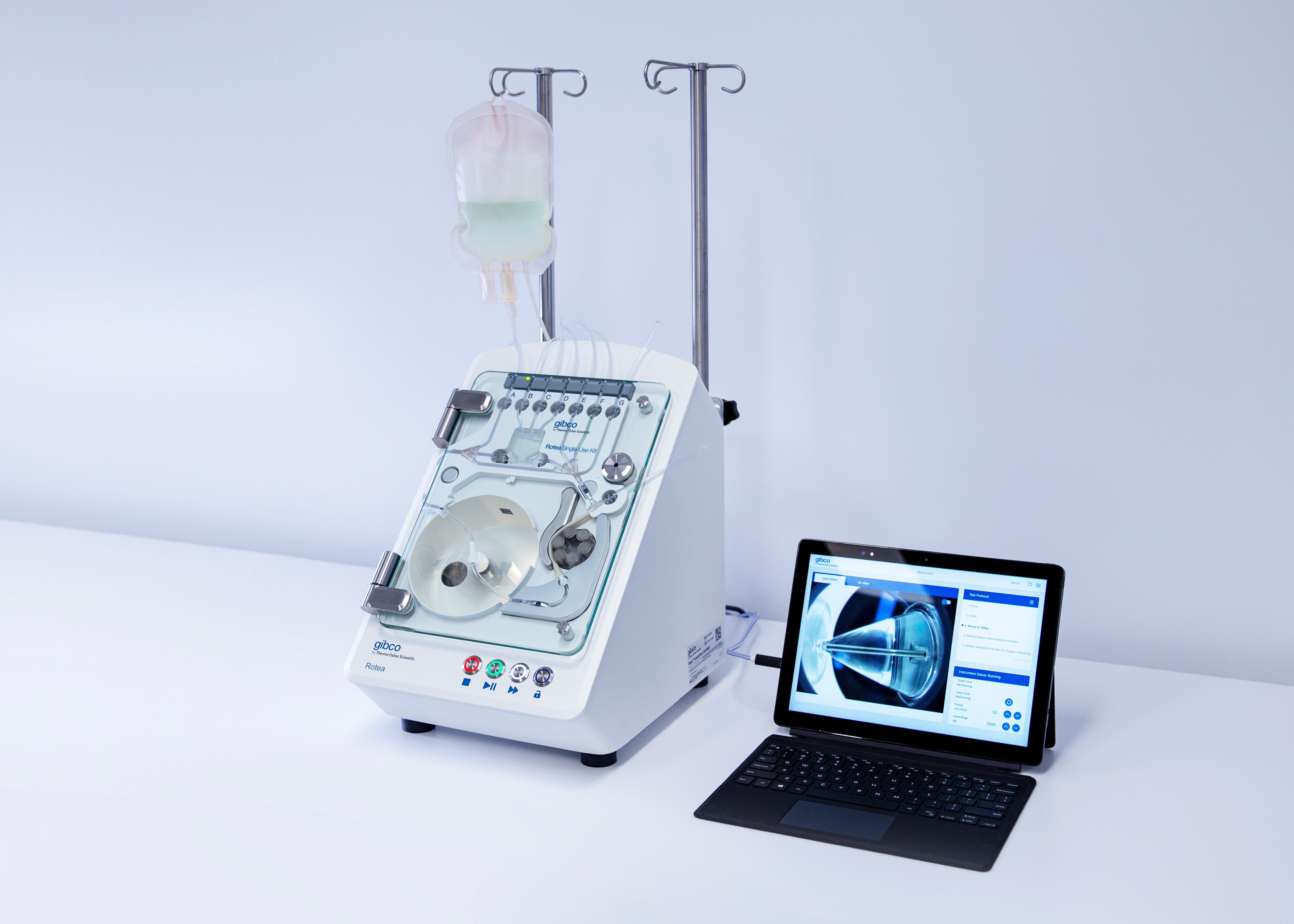
Scinogy’s multipurpose Gibco CTS Rotea Counterflow Centrifugation System has been configured, scaled and optimised for R&D and a seamless transition to commercial manufacture of high-quality, affordable cell and gene therapies, ultimately increasing patient access to these life-saving therapies.