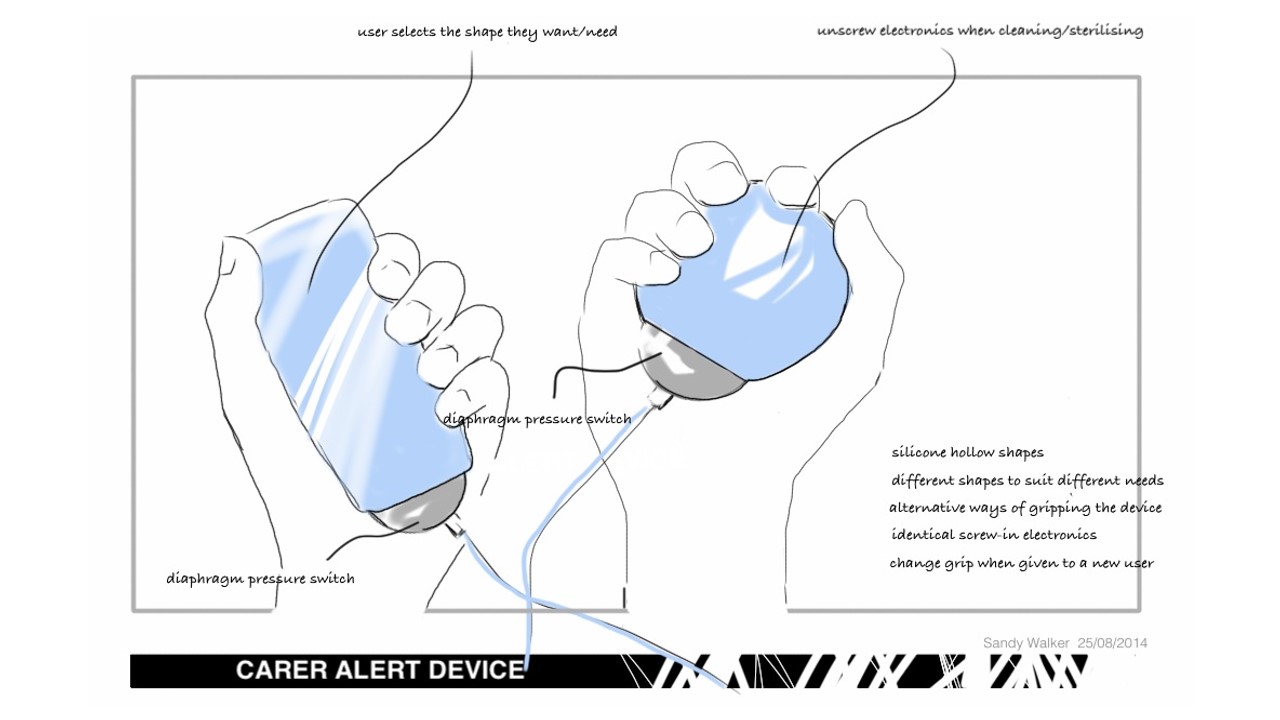
Arthritis Nurse Call Device

-
2016
-
Product
Medical and Scientific
Designed By:
A new nurse call assistive device suitable for the residential aged care market, with specific emphasis on making the device easier to use by residents who have manual dexterity problems and in particular, arthritis.