Optimized Positioning System OPS™

-
2015
-
Product
Medical and Scientific
Designed By:
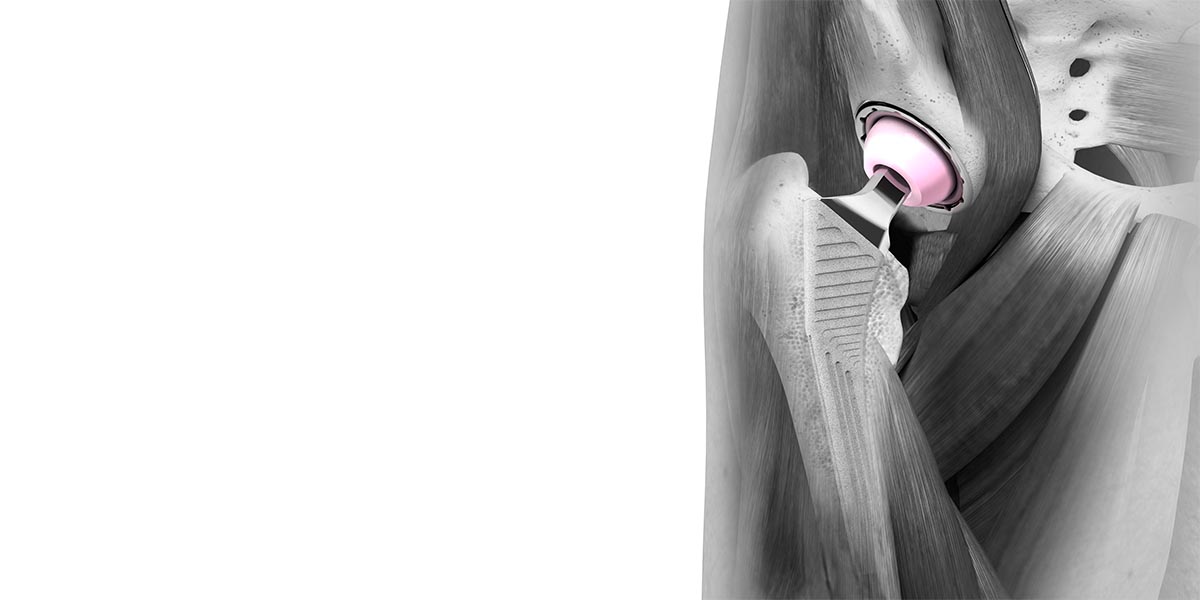
Every patient has a unique pelvic motion and physiological profile which can impact success of a Total Hip Replacement.
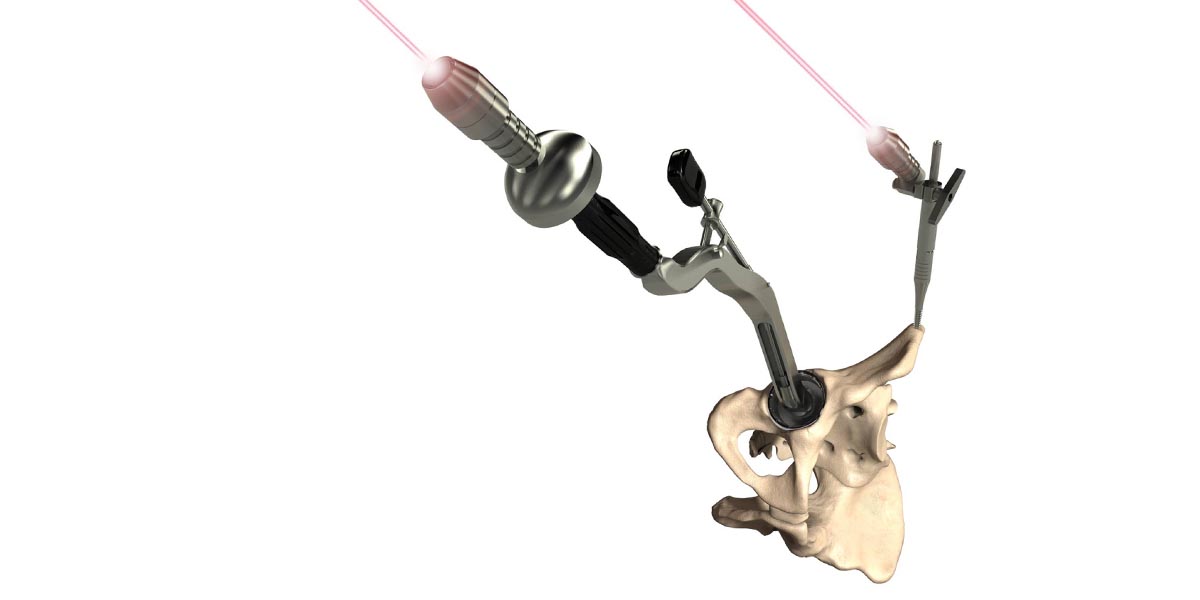
OPS™ is a two staged technology intended for use by orthopaedic surgeons to aid in the treatment of patients undergoing total hip replacement.
Stage 1 is a Dynamic Hip Analysis which provides information on the patient’s predicted biomechanics, displaying how the hip joint will be loaded post-operatively relative to varying orientations of the acetabular cup. The analysis is generated from patient X-ray and CT scans and delivered to the surgeon in a digital format.
Stage 2 is the Delivery System which enables accurate implant positioning in surgery using a 3D printed patient specific guide.